The Entire Process of Lipid Nanoparticle (LNP) Delivery for RNA Therapeutics + How to Design LNPs
The Entire Process of Lipid Nanoparticle (LNP) Delivery for RNA Therapeutics + How to Design LNPs!
Introduction
Lipid nanoparticles (LNPs) are lipid-based vesicles with a uniform lipid core, widely used for the delivery of small molecules and nucleic acid drugs. They have recently gained significant attention due to their remarkable success as the delivery platform for COVID-19 mRNA vaccines. The applications of transient protein expression induced by mRNA extend far beyond infectious disease vaccines, holding immense potential in areas such as cancer vaccines, protein replacement therapies, and gene editing components for rare genetic diseases. However, naked mRNA itself is highly unstable and prone to rapid degradation by nucleases and self-hydrolysis. Encapsulation within LNPs protects mRNA from extracellular RNases and assists in its intracellular delivery. In August 2021, Gaurav Sahay and colleagues published a review in Accounts of Chemical Research discussing the central role of LNPs in RNA delivery, offering valuable insights for LNP design and their broader applications.
Background
In recent years, lipid nanoparticles (LNPs) have successfully emerged as a delivery platform for RNA vaccines and therapeutics. Naked RNA is a negatively charged, hydrophilic macromolecule that struggles to enter cells due to electrostatic repulsion from the cell membrane and is rapidly degraded by ubiquitous RNases. Therefore, a protective carrier is necessary for cellular entry. Since cell membranes are primarily composed of lipids, utilizing lipid vesicles to encapsulate RNA facilitates passage through the cell membrane and release of the RNA into the cytoplasm. Consequently, these vesicles must initially consist of a positively charged lipid capable of binding the negatively charged RNA. However, vesicles composed of permanently cationic lipids can cause cytotoxicity due to electrostatic interactions with negatively charged cell membranes. Lipid structures have thus evolved into molecules that can become positively charged in response to the acidic environment of endosomes.
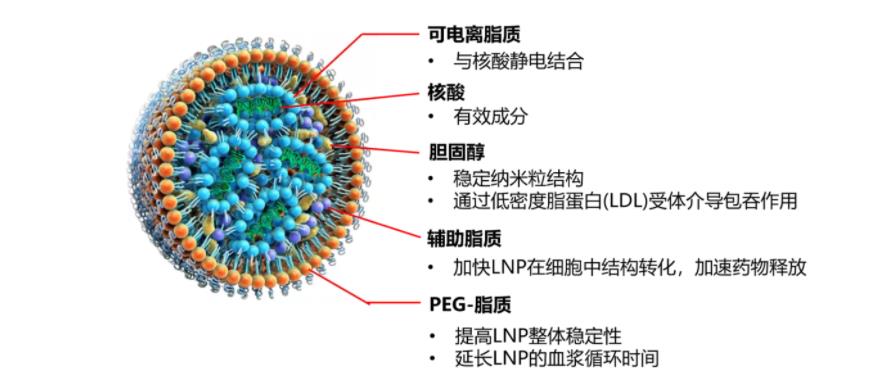
Figure 1: Structure of LNP
Currently, LNPs consist of structural lipids (which mimic the cell membrane and shield the positive charge) and polyethylene glycol (PEG)-modified lipids (which prevent LNP aggregation and unwanted interactions with the biological environment). The FDA approval of the first siRNA-LNP drug, Onpattro, the success of the mRNA-LNP COVID-19 vaccine (Comirnaty), and the Emergency Use Authorization (EUA) granted to Moderna's COVID-19 vaccine are landmark events in this field. LNP-based delivery platforms are safe for nucleic acid delivery and are equally suitable for delivering various therapeutic cargoes. Nevertheless, there is no universal solution applicable to all diseases, so ongoing optimization of LNPs continues.
LNP Preparation
The preparation of LNPs relies on their self-assembly capability, where lipid components spontaneously organize into nanostructured entities through intermolecular interactions. Initially, electrostatic binding occurs between the negatively charged nucleic acid and the positively charged lipid. Subsequently, assembly proceeds via hydrophobic interactions and van der Waals forces among the lipid components, ultimately forming the LNP. Due to the diversity of lipid chemistry, the unique characteristics of nucleic acids, and the temporal dynamics of their mixing, characterizing the early stages of self-assembly and understanding the impact on the final properties of LNPs remain challenging. The LNP preparation protocol influences the self-assembled product in at least two key aspects: LNP homogeneity and nucleic acid loading efficiency.
Numerous methods exist for LNP preparation, including liposome extrusion, thin-film hydration, nanoprecipitation, microfluidics, and impinging jet mixing. Among these, the rapid mixing of aqueous and lipid phases is a commonly used method. Microfluidics offers good reproducibility; however, its relatively low throughput makes it unsuitable for pilot-scale production. Pfizer utilized Knauer's IJM device, which employs impinging jet mixing. This equipment, characterized by high throughput and the absence of disposable consumables, proved highly effective in large-scale manufacturing.
LNP Formulation Components
Currently, the lipid composition of classic LNPs typically includes four components: an ionizable lipid (or cationic lipid), a helper lipid, cholesterol, and a PEGylated lipid (PEG-lipid).
Cationic Lipids (CLs) and Ionizable Lipids (ILs)
Cationic Lipids (CLs) and Ionizable Lipids (ILs) initiate the first step of self-assembly through electrostatic interactions. Lipid complexes containing cationic lipids are still widely used for nucleic acid delivery. However, due to toxicity concerns and lack of efficacy in vivo, they have largely been replaced by pH-responsive ionizable lipids. In LNP formulations, ionizable lipids are neutral at physiological pH but acquire a positive charge in the acidic environment of endosomes. This pH-dependent ionization makes ionizable lipids suitable materials for nucleic acid delivery, offering significantly improved efficacy and toxicity profiles. Ionizable lipids typically constitute 30%–50% of the total lipid in a formulation. Currently, much research is dedicated to fine-tuning the properties of ionizable lipids to further enhance efficiency, particularly for delivery to hard-to-reach human tissues.
The overall structure of both cationic and ionizable lipids can be divided into three parts: (1) the headgroup, (2) the linker, and (3) the tail.
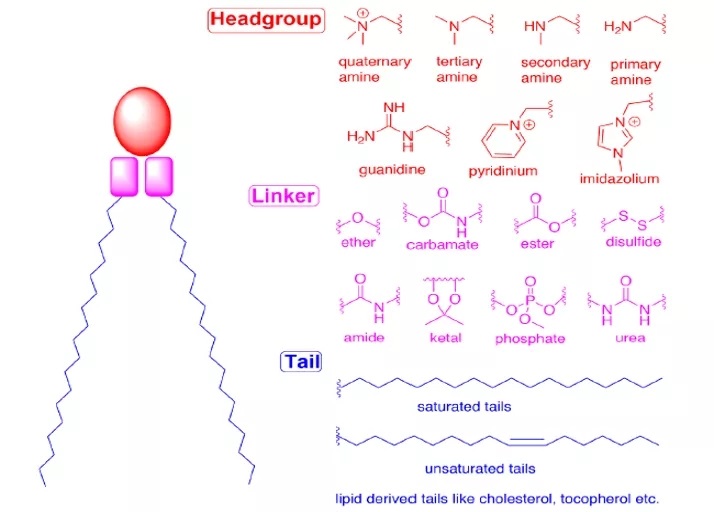
Figure 2: Overall Structure of CLs and ILs
Headgroup
The headgroup of ionizable lipids typically carries a positive charge. The size and charge density of the headgroup primarily participate in processes such as encapsulating nucleic acids, stabilizing the LNP, interacting with cell membranes, and promoting endosomal escape. Commonly used ionizable lipids contain only one headgroup, though sometimes they may contain several. Typical headgroups include amines (primary, secondary, tertiary, quaternary amines), guanidines, and heterocyclic groups, among others. The ionizable lipids used clinically (DLin-MC3-DMA, SM-102, ALC-0315) contain a tertiary amine headgroup, which undergoes pH-dependent ionization. The headgroups of ALC-0315 and SM-102 also contain a terminal hydroxyl group, which can reduce headgroup hydration and enhance hydrogen bonding interactions with nucleic acids, potentially improving transfection efficacy.
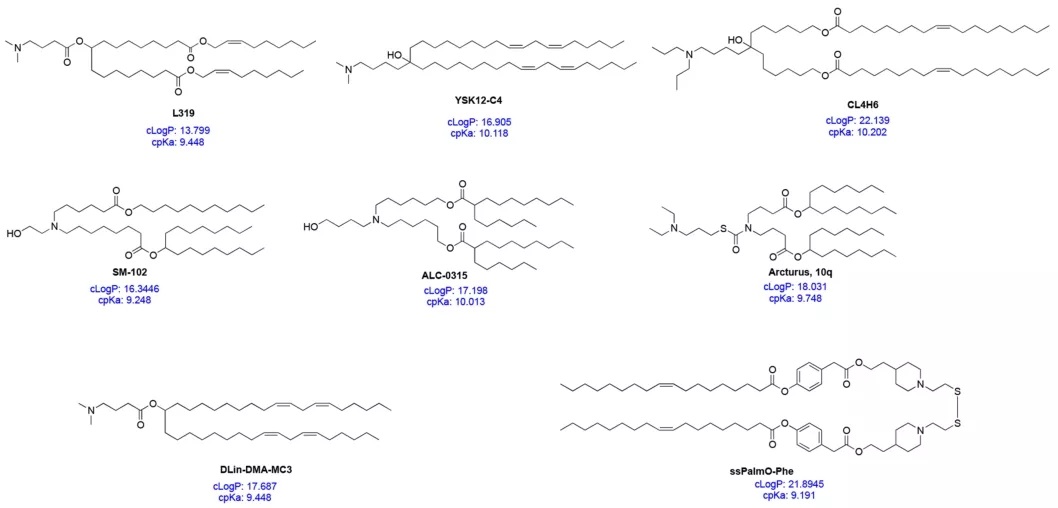
Figure 3: Structures, c*p*Ka, and cLogP of Selected ILs
Linker
The linker connects the headgroup to the tail(s); sometimes, the linker is embedded within the tail structure (e.g., in SM-102 and ALC-0315). The linker influences LNP stability, biodegradability, cytotoxicity, and transfection efficiency. Common linker types are shown in Figure 2. An ionizable lipid may contain one or multiple linkers; however, most ionizable lipids contain only one type of linker, likely due to ease of synthesis. Linkers can be categorized as non-biodegradable (e.g., ethers, carbamates) or biodegradable (e.g., esters, amides, thiols). Biodegradable linkers are often preferred because they typically enable rapid clearance in vivo, allow for multiple dosing, and reduce side effects. DLin-MC3-DMA, ALC-0315, and SM-102 all contain ester-based linkers. Modifications around the ester group in SM-102 can affect LNP clearance, formulation stability, and transfection efficiency.
Tail
The hydrophobic tail(s) influence pKa, lipophilicity, fluidity, and fusogenicity, thereby affecting LNP formation and potency. Ionizable lipids typically contain 1 to 4 hydrophobic tails, each comprising 8 to 20 carbon atoms. The tails can be saturated or unsaturated fatty acid chains. The degree of unsaturation can influence nucleic acid delivery by modulating membrane-destabilizing properties. DLin-MC3-DMA features two linoleyl tails, whereas ALC-0315 and SM-102 possess two branched, saturated tails, which are thought to adopt a conical geometry that promotes endosomal membrane destabilization and cytosolic release of the nucleic acid.
Properties of Ionizable Lipids
An ionizable lipid can be viewed as a multi-component molecule where each part must be carefully designed for the safe and efficient packaging and delivery of nucleic acids. Understanding the overall properties of ionizable lipids also aids in the development and design of next-generation candidates. The calculated pKa (c*p*Ka) is a key parameter, readily obtainable via computational methods. The c*p*Ka of ionizable lipids typically falls within the range of 9-10.5. Recent studies indicate that c*p*Ka closely approximates the actual pKa. The c*p*Ka of the ionizable lipid influences the overall pKa of the corresponding LNP formulation; when the ionizable lipid's c*p*Ka is around 8.5–10.5, the LNP's pKa is approximately 6–7. The difference between the ionizable lipid's c*p*Ka and the LNP's pKa appears consistent, around 2–4 units. Therefore, c*p*Ka can be used to guide the design of novel ionizable lipids.
Two other important property parameters for ionizable lipids are cLogP and cLogD, representing the lipophilicity of the molecule in its non-ionized and ionized states, respectively. Recent work by Rajappan et al. investigated the influence of pKa, c*p*Ka, and cLogD on the delivery efficiency of siRNA-LNPs, finding that ionizable lipids with a cLogD between 10 and 14 performed best. Given that common ionizable lipids have cLogP values in the range of 15–20, the lipophilicity of ionizable lipids should also be considered in the design of next-generation candidates. Since the ionization property (c*p*Ka) and lipophilicity (cLogP) of an ionizable lipid can influence processes ranging from initial complex formation with the nucleic acid to final nanoparticle formation and cargo delivery, considering both c*p*Ka and cLogP simultaneously could accelerate the development of effective ionizable lipids. However, more studies are needed to further substantiate this conclusion.
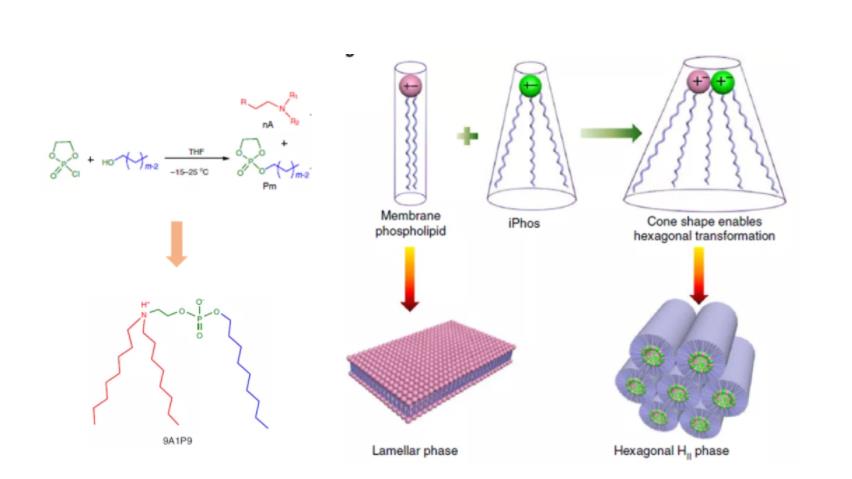
Figure 4: iPhos Amphoteric Lipids
In addition to traditional cationic lipids and ionizable lipids, there also exist amphoteric lipids. In a recent study, Liu et al. synthesized a library of over 500 amphoteric lipid molecules, termed iPhos. iPhos consists of an amine group, a short hydrophobic tail, and a phosphate group serving as the linker. It is proposed that the negatively charged phosphate group promotes membrane fusion and induces endosomal escape. Among these, formulation 9A1P9 was found to preferentially deliver target nucleic acids to the liver and lungs.
In summary, the properties of each component of an ionizable lipid can influence the overall formulation and biological characteristics of the LNPs. Over the past 50 years, extensive systematic research has been dedicated to designing ideal ionizable lipids. Some of these have been approved by the FDA for the delivery of genetic cargo. However, significant further research is needed to design non-toxic, non-hepatotropic ionizable lipids capable of efficiently delivering different types of genetic cargo.
■ Cholesterol
Cholesterol, a naturally abundant component of cell membranes, is commonly used as a structural lipid in LNP formulations, typically constituting about 20-50% of the lipid composition. It is a plentiful component of animal cell membranes and is usually extracted in large quantities from natural sources like wool. The role of cholesterol in cellular uptake was overlooked for a long time. Studies have found that replacing cholesterol in LNP formulations with natural phytosterols (such as β-sitosterol) or oxidized cholesterol derivatives can significantly improve mRNA delivery, likely due to altered lipid trafficking in the endocytic pathway and enhanced endosomal escape of the LNPs. Although it is not yet fully understood why LNPs containing β-sitosterol are more likely to avoid the lysosomal pathway, their shape polymorphism and multilamellar structural organization might deform the endosomal membrane or extend the timeframe for nucleic acid release. Cholesterol is primarily located in the outer shell of the LNP, which explains why modifications to the sterol structure might induce organizational changes on the LNP surface.
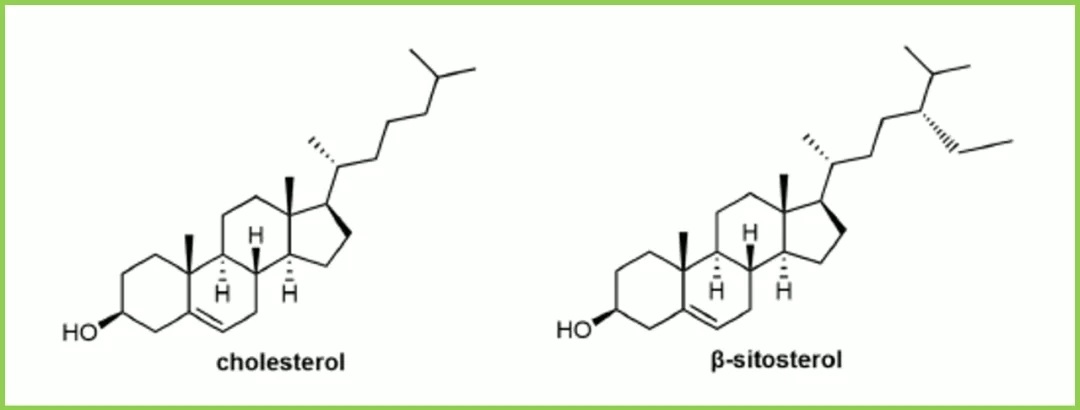
Figure 5: Cholesterol and β-Sitosterol
Furthermore, recent studies indicate that cholesterol relocates from the LNP core to the lipid shell upon interaction with apolipoprotein E (ApoE). These findings suggest that although cholesterol is relatively inert compared to ionizable lipids, it and its derivatives can influence cellular recognition pathways. However, numerous unknowns and challenges remain regarding the modification of cholesterol in LNP formulations. For instance, phytosterols are not as abundant as cholesterol; their scarce supply and high production costs may hinder their application in LNP formulations. The nature of the raw materials and corresponding trace impurities could lead to batch-to-batch variability in LNPs. The stereochemistry of sterols and lipids may also potentially impact LNP formulations. For example, does stereochemistry affect lipid recognition, trafficking, and recycling? Are there significant differences in lipid packing at the LNP surface between different enantiomers? Answers to these questions require further investigation.
■ Phospholipids
Phospholipids contribute to encapsulating nucleic acids and stabilizing the LNP. Compared to other components, they have been relatively less studied and typically constitute 10%–20% of the total lipid in a formulation. Phospholipids often serve as structural lipids in LNP formulations because they spontaneously organize into lipid bilayers, and their higher phase transition temperatures enhance the membrane stability of the LNPs. Similar to cell membranes, phospholipids are located at the periphery of the LNP. These lipids are usually semi-synthetic. For example, phosphatidylcholine is commonly sourced from natural raw materials like egg yolk and soybeans and can be chemically modified (e.g., by extending the fatty acid tails).
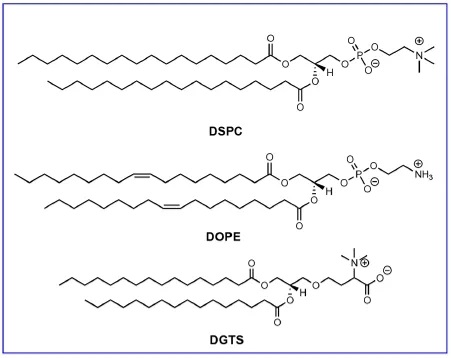
Figure 6: Structures of DSPE, DOPE, and DGTS
1,2-distearoyl-sn-glycero-3-phosphocholine (DSPC) is a structural lipid used clinically in LNPs for the siRNA therapeutic Onpattro and SARS-CoV-2 mRNA vaccines. The structure of DSPC consists of a phosphatidylcholine headgroup and two saturated 18-carbon tails. These two tails form a tightly packed lipid bilayer. In LNPs, DSPC is primarily located at the nanoparticle surface and in more peripheral positions within the nanoparticle core. 1,2-dioleoyl-sn-glycero-3-phosphoethanolamine (DOPE) is another phospholipid commonly used in preclinical LNP research. The unsaturated tails of DOPE not only form a more fluid lipid layer but also enable the formation of a hexagonal (HII) phase organization. The HII phase can promote fusion between the lipid membrane and the endosomal membrane, leading to the cytosolic release of nucleic acids.
Some studies have shown that DOPE-LNPs can enhance RNA transfection efficiency compared to DSPC-LNPs. Recently, Zhang et al. reported that after intravenous administration, C12-200 LNPs containing DOPE accumulated in the liver, whereas C12-200 LNPs containing DSPC accumulated in the spleen, demonstrating the influence of the structural lipid on LNP biodistribution. Sahay et al. found that in MC3-LNPs, replacing DSPC with natural glycolipids affected mRNA transfection efficiency. The plant-derived membrane lipid 1,2-dipalmitoyl-sn-glycero-3-O-4'-(N,N,N-trimethyl)-homoserine (DGTS) exhibited varying transfection efficiencies depending on the administration route. In summary, these studies indicate that structural lipids play a significant role in LNP-based RNA delivery.
■ PEG-Modified Lipids
PEG-modified lipids (PEG-lipids) are an important component in LNPs for modulating half-life and cellular uptake. During LNP assembly, due to their hydrophilicity and large size, PEG chains reside on the outer shell of the nanoparticle. As with other nanocarriers, PEG provides an external polymer layer for LNPs that hinders serum protein adsorption and uptake by the mononuclear phagocyte system, thereby prolonging their circulation time in vivo. PEG also prevents nanoparticle aggregation during storage and in the bloodstream. Furthermore, the amount of PEG-lipid can determine the particle size. Another potential purpose of PEG-lipids is to enable LNP surface functionalization. Functionalized PEG-lipids can be bioconjugated to ligands or biomacromolecules. For example, Singh et al. used DSPE-PEG-amine conjugated to hyaluronic acid for tumor targeting; Parhiz et al. employed DSPE-PEG-maleimide for antibody conjugation.
Although PEG is crucial for LNP stability and bioconjugation, its desorption is also critical for cell transfection. The desorption (or shedding) of PEG from the LNP allows opsonization by serum proteins, such as apolipoproteins and albumin, which are key effectors for receptor-mediated endocytosis of LNPs. Akinc et al. demonstrated that ApoE binding to LNPs enables low-density lipoprotein receptor (LDLR)-mediated hepatocyte internalization.
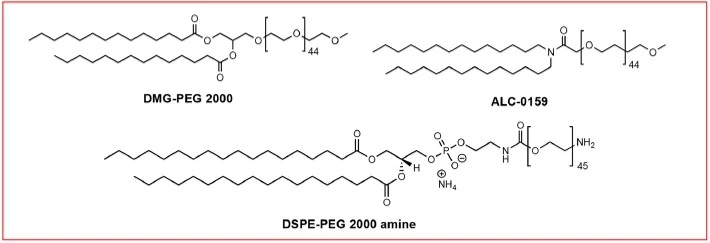
Figure 7: PEG-Lipid Structures
Because PEG-lipids can inhibit the binding of ApoE to LNPs, an excessive amount of PEG-lipid may adversely affect LNP cellular uptake and transfection. LNPs containing less PEG-lipid appear to deliver nucleic acids more efficiently, likely due to easier association with ApoE. The lipid chain length of the PEG-lipid is also a crucial factor determining its desorption rate. Mui et al. reported that the extent of PEG dissociation from the LNP membrane is inversely related to the length of the lipid chain in the PEG-lipid, as the hydrophobic interaction between the PEG-lipid and the LNP membrane increases with longer PEG-lipid lipid chains.
Suzuki et al. suggested that the rate of PEG shedding might also influence the generation of anti-PEG antibodies, potentially causing complications upon repeated dosing. In LNP formulations for intravenous injection, the PEG-lipid content rarely exceeds 2%. However, a dense PEG layer might be beneficial for achieving extrahepatic targeting. Studies by Lee et al. showed that LNPs containing 5% PEG-lipid accumulated in tumors to a greater extent than those containing 2.5% PEG-lipid. Lokugamage et al. demonstrated that PEG-lipids are crucial for the delivery of nebulized LNPs. Therefore, the amount and type of PEG-lipid in LNPs likely need careful adjustment based on clinical requirements.
LNP Properties
The average particle size and size distribution of LNPs are important initial determinants of LNP quality and suitability for various applications. These characteristics are typically characterized by dynamic light scattering (DLS). Generally, the optimal size range for LNPs is 20–200 nm, as this size makes LNPs robust enough to withstand fluid flow (e.g., in blood and lymph) while allowing them to penetrate interstitial spaces. LNP size is often modulated by altering the amount of PEG-lipid or mixing parameters (such as flow rate and volume ratio). LNP size can influence their internalization, biodistribution, degradation, and clearance, and different applications may require different particle sizes. For example, in mouse models, 45 nm siRNA-LNPs showed the best delivery efficiency upon subcutaneous administration, whereas 80 nm siRNA-LNPs were most effective upon intravenous administration. However, comparisons of different mRNA-LNP sizes in rodents and non-human primates suggested that size is less critical in non-human primates when LNPs are administered intramuscularly.
The surface charge of LNPs governs their interactions with cell membranes and the biological environment. Because cell membranes are negatively charged, LNPs with a negative surface charge experience repulsion and are not readily taken up by cells. On the other hand, positively charged LNPs might directly disrupt cell membranes, causing cytotoxicity. This is why ionizable lipids are crucial in LNP design: initially, LNPs containing ionizable lipids are electrically neutral, avoiding unwanted electrostatic interactions, but they acquire a positive charge in the acidic pH of endosomes. The surface charge of LNPs is typically assessed by measuring the Zeta potential. This technique is commonly used to evaluate colloidal aggregation. While there is no strict classification, a surface charge is generally considered weak if the zeta potential is between −20 and +20 mV.
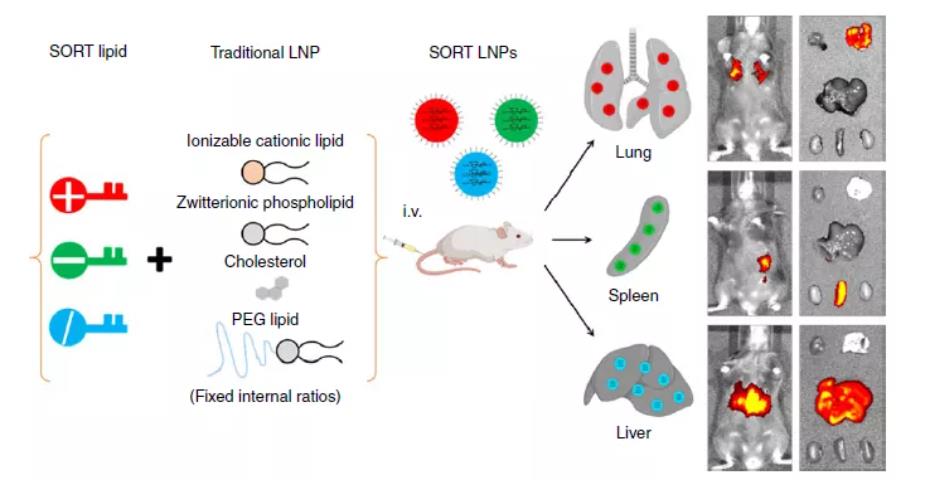
Figure 8: SORT-LNP
A common method for modulating the total surface charge of LNPs is by adjusting the N/P ratio, which is the ratio of ionizable lipid (N, representing cationic amine groups) to nucleic acid (P, representing anionic phosphate groups). Carrasco et al. reported that in LNPs containing the ionizable lipid KC2, increasing the N/P ratio increased both the surface charge and the encapsulation efficiency. Furthermore, incorporating permanently charged lipids into LNPs can alter the preferential uptake by organs without necessarily increasing the net surface charge. Cheng et al. demonstrated Selective Organ Targeting (SORT) in mice based on lipid charge: adding a positively charged lipid to the LNP formulation directed preferential transfection to lung tissue, while a negatively charged lipid directed preferential transfection to the spleen.
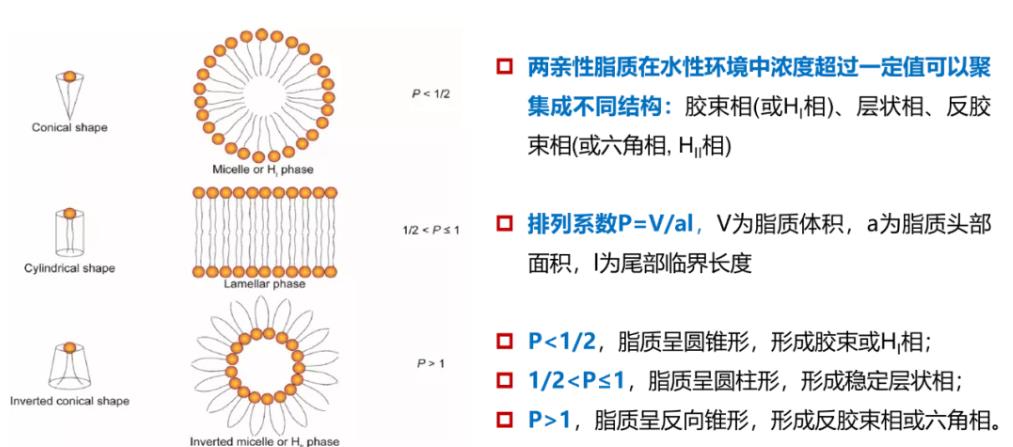
Figure 9: Lipid Shape and Structure
Lipid packing can influence numerous parameters, ranging from membrane hydration and deformability to cellular uptake and cargo release. In brief, each lipid can be described by a packing parameter determined by the volume ratios of its polar "head" and nonpolar "tail." Structurally uniform lipids tend to form cylindrical structures and lamellar phases, whereas non-uniform lipids form hexagonal, cubic, and micellar phases. The inverse hexagonal phase (HII) appears to significantly promote lipid membrane fusion. Currently, in the field of RNA-LNP delivery, the controlled formation of non-lamellar phases, such as cubic phases, remains relatively uncommon. However, LNPs may undergo structural changes when exposed to environmental triggers. Heyes et al. utilized ³¹P NMR spectroscopy to investigate the phase transition behavior of lipid particles containing a series of cationic lipids with different lipid tails. They found that lipids with a lower lamellar-to-hexagonal transition temperature (T<sub>BH</sub>) more readily undergo membrane fusion, a result ultimately corroborated by gene silencing efficiency.
Similarly, the membrane destabilization theory proposes that when ionizable lipids are exposed to the acidic pH environment of late endosomes, electrostatic interactions between the ionizable lipids and phospholipids in the endosomal membrane lead to membrane disruption. A recent study by Liu et al. applied this concept, reporting evidence—shown by ³¹P NMR spectroscopy—of hexagonal phase formation when a novel ionizable lipid was exposed to a simulated endosomal environment. Although pH-induced binding is the most common mechanism for lipid materials, other methods inducing endosomal membrane destabilization have also been reported.

Figure 10: Membrane Destabilization Achieved by Ionizable Lipids
Depending on the lipid phase and overall polarity, lipid membranes may entrap water molecules, thereby altering membrane fluidity or deformability, which subsequently affects membrane fusion. Membrane hydration can also influence potential responsiveness to pH—a key environmental trigger commonly utilized to initiate nucleic acid release. The pH variations encountered by LNPs throughout their lifecycle are illustrated in Figure 11. When LNPs enter the intracellular space, they become trapped in endosomes, which gradually acidify as they mature into lysosomes. Consequently, higher water content within the lipid membrane may impact acidification kinetics and contribute to rapid membrane destabilization. Koitabashi et al. employed Laurdan assay to examine the pH-responsive destabilization of lipid membranes in siRNA-LNPs and found a positive correlation between membrane hydration and gene silencing efficiency.
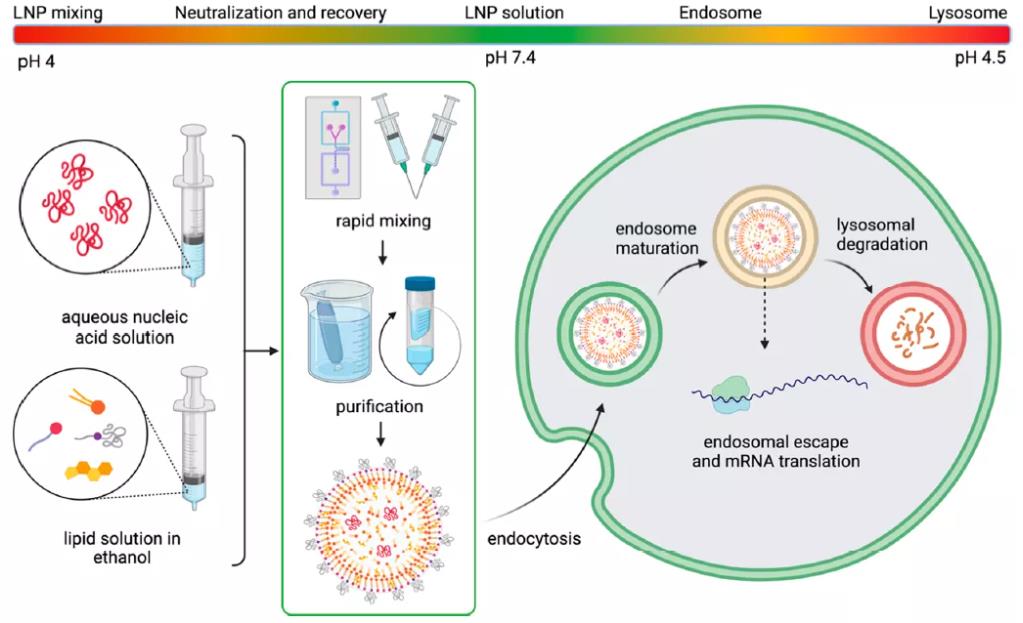
Figure 11: pH Changes Encountered by LNPs During Their Lifecycle
An interesting observation regarding membrane hydration is that ³¹P NMR experiments demonstrated siRNA-LNPs exhibit lower water content compared to mRNA-LNPs of the same formulation, likely due to the longer length of the hydrophilic mRNA strands. These findings further support the view of Carrasco et al., who discovered that KC2-LNPs with lower N/P ratios contain more mRNA and lipid per nanoparticle and exhibit a higher dielectric constant, indicating greater hydration in low N/P ratio LNPs compared to high N/P ratio LNPs. Enhanced RNA solvation further improves transfection efficiency. mRNA-LNPs may be more sensitive to environmental changes, although variations in pH sensitivity may not align with the timescale of biological processes. The reorganization of LNPs upon exposure to the biological environment further complicates the issue of shell hydration.
The inherent aqueous environment of LNPs also poses a threat to their long-term stability. Under normal ambient conditions, pure nucleic acids can degrade rapidly via exogenous RNase degradation or self-hydrolysis. While LNPs protect nucleic acids from enzymatic degradation, thermodynamic factors (such as minimizing phase separation) cause LNPs to tend to aggregate, leading to nucleic acid loss during nanoparticle fusion and ultimately affecting transfection efficiency. Low-temperature storage and lyophilization may preserve the RNA but can disrupt LNPs due to ice crystal formation; adding cryoprotectants like sucrose appears to mitigate this issue. Interestingly, LNPs may alter their organ targeting preferences based on storage conditions, likely due to structural reorganization.
It is noteworthy that the storage requirements differ between the Pfizer/BioNTech and Moderna vaccines, suggesting that variations in LNP formulations can significantly alter nucleic acid affinity and LNP stability. Currently, there is no established method to accelerate stability testing for these non-crystalline materials, meaning stability must be assessed empirically at discrete time points. Thermal methods such as differential scanning calorimetry (DSC) may provide valuable assays for LNP degradation. Ultimately, a better understanding of LNP self-assembly is required to predict the downstream properties of LNP-based RNA therapeutics.
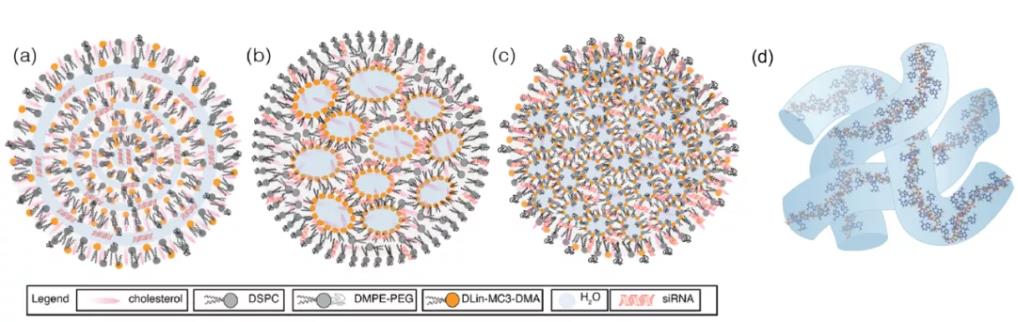
Figure 12: Structure of siRNA-LNP. (a) Multilamellar vesicle; (b) Nanostructured core; (c) Homogeneous core-shell; (d) mRNA aqueous column
The internal organizational structure of LNPs remains an unresolved issue. Some reports suggest that siRNA-LNPs possess a "sandwich" structure where the siRNA interacts with the outer lipid layers of the LNP. On the other hand, mRNA may also be stretched along the LNP periphery, occupying the water-lipid interface inside "vesicular" LNPs, or residing within interconnected aqueous channels where the mRNA might be displaced due to its hydrophilicity. The mRNA could also be sequestered from the bulk lipid into aqueous compartments. Recently, cryo-TEM studies by Sahay et al. indicated that any deviation from uniform, spherical LNPs (e.g., polymorphic shapes, multilamellar structures, or vesicles) might enhance mRNA transfection efficiency. However, it remains unclear whether the improved transfection is influenced by molecular recognition of the lipid components.
Variations in lipid organization may also be affected by solubility. Although lipids are typically dissolved before mixing via heating or sonication, Yanez Artla et al. reported detecting cholesterol crystals within the LNP lipid membrane using small-angle X-ray scattering. Lipid organization can also change upon exposure to the biological environment; when exposed to the abundant serum protein ApoE, LNPs undergo reorganization in both their surface and core. The unresolved questions surrounding lipid organization in RNA delivery require further investigation.
Nanoparticles are prone to nonspecific adsorption of proteins, forming a biomolecular corona at the interface. The formation of this biomolecular corona on the nanoparticle surface alters its surface properties and physicochemical characteristics, playing a significant role in the nanoparticle's biodistribution and phagocytosis. ApoE is a component of this corona and influences LNP-mediated nucleic acid delivery. Primarily synthesized by hepatocytes and abundant in the liver, ApoE partially explains why intravenously injected LNPs typically accumulate in the liver. ApoE is intrinsically involved in cholesterol metabolism by forming lipid complexes and transporting them to cells expressing the low-density lipoprotein receptor (LDLR), promoting LDLR-mediated endocytosis. This concept also applies to LNPs; Akinc et al. demonstrated significantly reduced cellular uptake of LNPs containing the ionizable lipid KC2 under conditions lacking ApoE or LDLR compared to normal conditions. Dong reported that LNPs containing cKK-E12 (an ionizable lipid) enter cells more efficiently in the presence of ApoE.
Other lipids in the LNP formulation may also be involved in ApoE binding to LNPs. Zhang et al. reported that, with other lipid components being identical, DOPE interacts more strongly with ApoE than DSPC, indicating that changes in phospholipid structure alter the binding affinity of ApoE for LNPs. Sebastiani et al. reported that the adsorption of ApoE onto mRNA-LNPs induces a rearrangement of the lipid components within the nanoparticle, relocating some cholesterol from the nanoparticle core to the shell upon exposure to ApoE. Kim et al. reported that altering PEG content and conjugating PEG-lipids to mannose could control cell-specific delivery of mRNA within the liver. Interestingly, ApoE is also abundant in the brain and central nervous system, where astrocytes can produce it. Tanaka et al. explored delivering mRNA-LNPs to mice via intracerebroventricular injection and found successful uptake of ssPalm-LNPs by astrocytes.
ApoE is not the only protein known to be associated with differences in cellular uptake. Recently, Miao et al. demonstrated that serum albumin allows LNPs to enter cells via an ApoE-independent pathway, highlighting the importance of other serum proteins. Despite numerous findings, how to fully leverage the potential of the biomolecular corona remains unclear. The biomolecular corona governs immune system recognition of LNPs, circulation time, and biodistribution. Finding methods to control the corona's composition could enable cell-, tissue-, or organ-specific uptake.
Summary
LNPs are highly customizable nucleic acid delivery vehicles that have demonstrated immense potential in mRNA vaccine delivery. Furthermore, their potential value in treating rare diseases and cancer should not be overlooked. mRNA therapies can facilitate the production of therapeutic proteins to restore function in damaged tissues or organs. Substantial research efforts worldwide are dedicated to designing and refining the individual components of LNPs for the efficient and safe delivery of target nucleic acids.
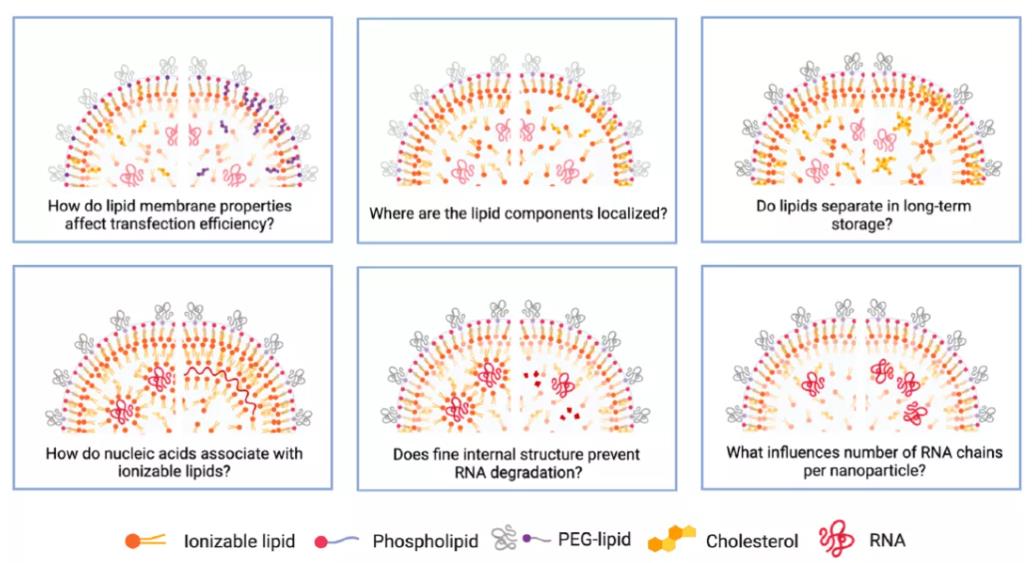
Figure 13: Unresolved Questions Regarding LNPs
However, numerous questions about LNPs remain unanswered. For example:
How do the properties of the lipid membrane influence transfection efficiency?
What is the precise localization of each lipid component within the LNP?
Does lipid segregation occur during long-term storage?
How do nucleic acids interact with ionizable lipids?
How does the internal structure of LNPs protect nucleic acids from degradation?
What factors determine the number of RNA molecules per LNP?
With the surge in public attention on mRNA vaccines, related research is expected to advance rapidly, potentially ushering in a new era of nanomedicine.
References:
Yulia Eygeris, Mohit Gupta, Jeonghwan Kim, Gaurav Sahay. Chemistry of Lipid Nanoparticles for RNA Delivery. Acc. Chem. Res. 2022, 55, 2-12.
Yuebao Zhang, Changzhen Sun, Chang Wang, Katarina E. Jankovic, and Yizhou Dong. Lipids and Lipid Derivatives for RNA Delivery. Chem. Rev. 2021, 121, 12181–12277.
Linde Schoenmaker, Dominik Witzigmann, Jayesh A. Kulkarni, Rein Verbeke, Gideon Kersten, Wim Jiskoot, Daan J.A. Crommelin. mRNA-lipid nanoparticle COVID-19 vaccines: Structure and stability. Int. J. Pharm. 2021, 601, 120586.