How are the lipid nanoparticles (LNPs) in mRNA COVID-19 vaccines assembled?
Based on the development of mRNA COVID-19 vaccines, lipid nanoparticles (LNPs) have been proven to be an effective delivery method for RNA-based drugs and vaccines. LNPs encapsulate and protect degradable active ingredients, mimic low-density lipoproteins (LDLs), and are taken up via endogenous pathways. Designed to be pH-sensitive, LNPs release their payload into the cytoplasm. This marks the first large-scale use of nucleic acid-loaded lipid nanoparticles in the history of vaccination.
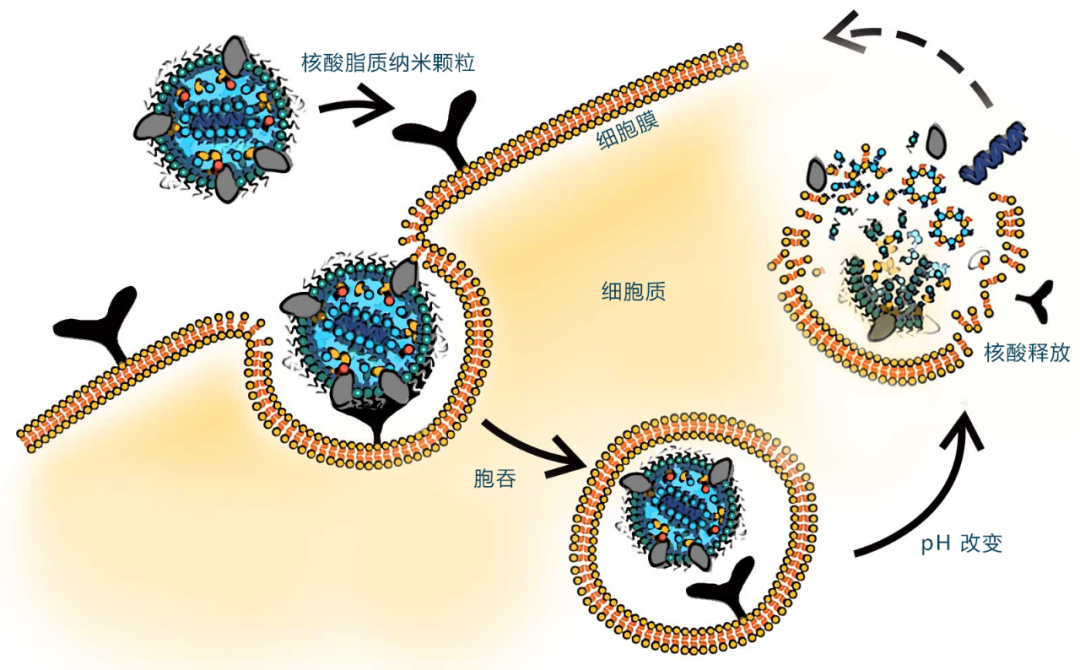
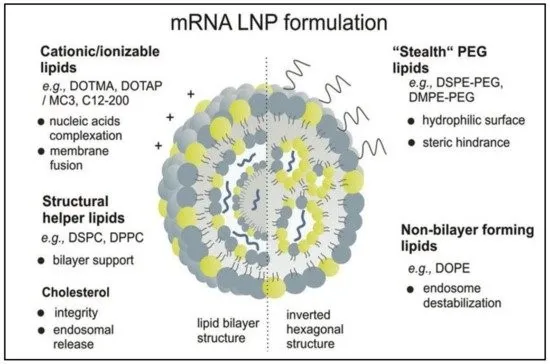
The structures of currently approved mRNA vaccines are largely similar (with different companies employing distinct molecular structures and ratio formulations):
① mRNA – the active ingredient responsible for the vaccine's effect;
② Cationic lipid – binds to negatively charged mRNA and serves as a key structural component;
③ Cholesterol – mediates LNP endocytosis and stabilizes the LNP structure;
④ Phosphatidylcholine – an auxiliary lipid that facilitates faster mRNA release during endocytosis;
⑤ PEGylated phospholipid – extends metabolic half-life and enhances LNP stability.
So the question arises: how are these structurally complex LNPs assembled?
This is perhaps the most significant challenge in mRNA vaccine production.
How does one control the composition, particle size, flow rate, morphology, and other parameters of LNPs while ensuring quality and accelerating production?
Literature reports various assembly methods, such as the thin-film method and T-junction mixing. However, the issue is that these methods have low throughput and are difficult to use for large-scale production.
To overcome these challenges, BioNTech/Pfizer employed an impinging jet mixing method. Using a BlueShadow 80P high-pressure pump, they directed two jets – one containing the API (mRNA) and the other the lipid solution – to collide within a chamber. This utilizes fluid dynamics to ensure thorough mixing of all LNP components, forming the lipid nanoparticles that encapsulate the mRNA.

Figure: IJM Impinging Jet Mixer
The Impinging Jet Mixer (IJM) required for LNP production equipment was developed and manufactured by the German laboratory instrument manufacturer KNAUER. The IJM is designed and optimized to meet customer performance requirements and regulatory standards. It has been successfully installed and accepted within a Grade C cleanroom. The system is equipped with all necessary interfaces for integration into a PLC system. A central control platform manages the flow rates of all units within the process step. All units are mounted within a stainless-steel frame to accommodate Clean-in-Place (CIP) procedures in pharmaceutical production.
The system includes:
KNAUER Impinging Jet Mixer (IJM)
Inlet lines for the lipid/ethanol mixture and the mRNA/buffer mixture
LNP outlet line
Cleaning system
Frame (stainless steel, grade 1.4301)
Technical documentation from KNAUER, including installation and training guides, service documents, and GMP files
Software system compliant with regulatory standards such as 21 CFR Part 11
Factory Acceptance Test (FAT)
Site Acceptance Test (SAT)